 Clinical trials have demonstrated the effectiveness of NeuroStar® TMS Therapy in treating patients who have not benefited from prior antidepressant medication. NeuroStar TMS Therapy was studied in adult patients suffering from Major Depressive Disorder, all of whom had not received satisfactory improvement with previous treatments.
Clinical trials have demonstrated the effectiveness of NeuroStar® TMS Therapy in treating patients who have not benefited from prior antidepressant medication. NeuroStar TMS Therapy was studied in adult patients suffering from Major Depressive Disorder, all of whom had not received satisfactory improvement with previous treatments.
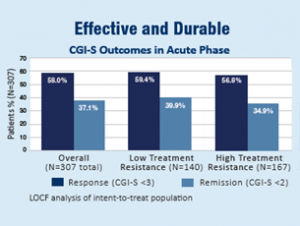
An Effective and Durable Option for Treating Major Depressive Disorder
In an independent, randomized, controlled trial funded by the National Institute of Mental Health, 307 patients were treated with the NeuroStar TMS System for 4 to 6 weeks, similar to real clinical context.1 Patients were divided into two groups:
- Low Treatment Resistance: Patients who have failed to improve their depression symptoms after a single antidepressant treatment of adequate dose and duration.
- High Treatment Resistance: Patients who have failed to improve their depression symptoms after a multiple (2-14) antidepressant treatments of adequate dose and duration.
Patients treated with the NeuroStar TMS System also experienced significant improvement in anxiety and physical symptoms (such as appetite changes, aches and pains, and lack of energy) associated with depression.1
Durability of TMS Treatments
Treatment Algorithm
Additionally, Neuronetics has developed a new tool to use in educating patients on the best practices of treating depression. The Best Practices Treatment Guideline for Depression has been developed to help patients understand TMS therapy as an option if their first line antidepressant medications stop working. This guideline is based on the 2010 American Psychiatric Association’s practice guidelines and NeuroStar TMS Therapy indication for use, which says: NeuroStar TMS Therapy is indicated for the treatment of major depressive disorder in adult patients who have failed to achieve satisfactory improvement from prior antidepressant medication at or above the minimal effective dose and duration in the current episode. Please see the Treatment Algorithm for an effective illustration of the use of NeuroStar TMS Therapy early on in the treatment of depression. NeuroStar TMS Therapy has not been studied in patients who have not received prior antidepressant treatment. View TMS Therapy safety data, TMS+YOU is an online community and national patient advocacy site for TMS therapy.
Those considering Ttranscranial magnetic stimulation can connect with patients who have had the treatment to answer questions, share insights, and get the latest information.
References:
Carpenter LL, et al. (2012). Depress Anxiety, 29(7):587-596.
